Quality control of ultrasonic elements
TRZ® Analyzer applications
Manufacturing high quality industrial or medical ultrasonic parts is not an easy task, even with a good design, since the performance of these elements is sensitive to many variables. For example, any changes in finish, dimensions, material properties and manufacturing procedure will affect frequency, impedance, and mechanical quality factor. This high sensitivity is responsible for the large standard deviation and number of warranty failures observed in the routine of many manufacturers of power ultrasonic equipment. Failures are especially critical in the case of surgical equipment. These considerations are valid for transducers, converters, acoustic amplifiers, waveguides, sonotrodes and inserts.
The quality control of ultrasonic parts must ensure that frequencies, impedances, and the mechanical quality factor are within pre-established acceptance ranges. It is also necessary to record and document the results for traceability and to use calibrated test instruments.
The TRZ® Analyzer has been continuously improved with each generation and features dedicated resources for quality control, such as programmable acceptance criteria, PDF report generation, and data export to spreadsheets. It also evaluate presents results in green (pass) and red (fail) colors to facilitate use in the production line.
Next, the TRZ® Software interface for information on measurement parameters and programmable judgment criteria is shown. It is possible to preset and customize a total of 30 sets of parameters and criteria. The parameters consist of the sweep start and stop frequency, the number of points and the refinement option. The criteria consist of acceptance ranges for resonance and anti-resonance frequencies, impedance at resonance and anti-resonance, and mechanical quality factor.
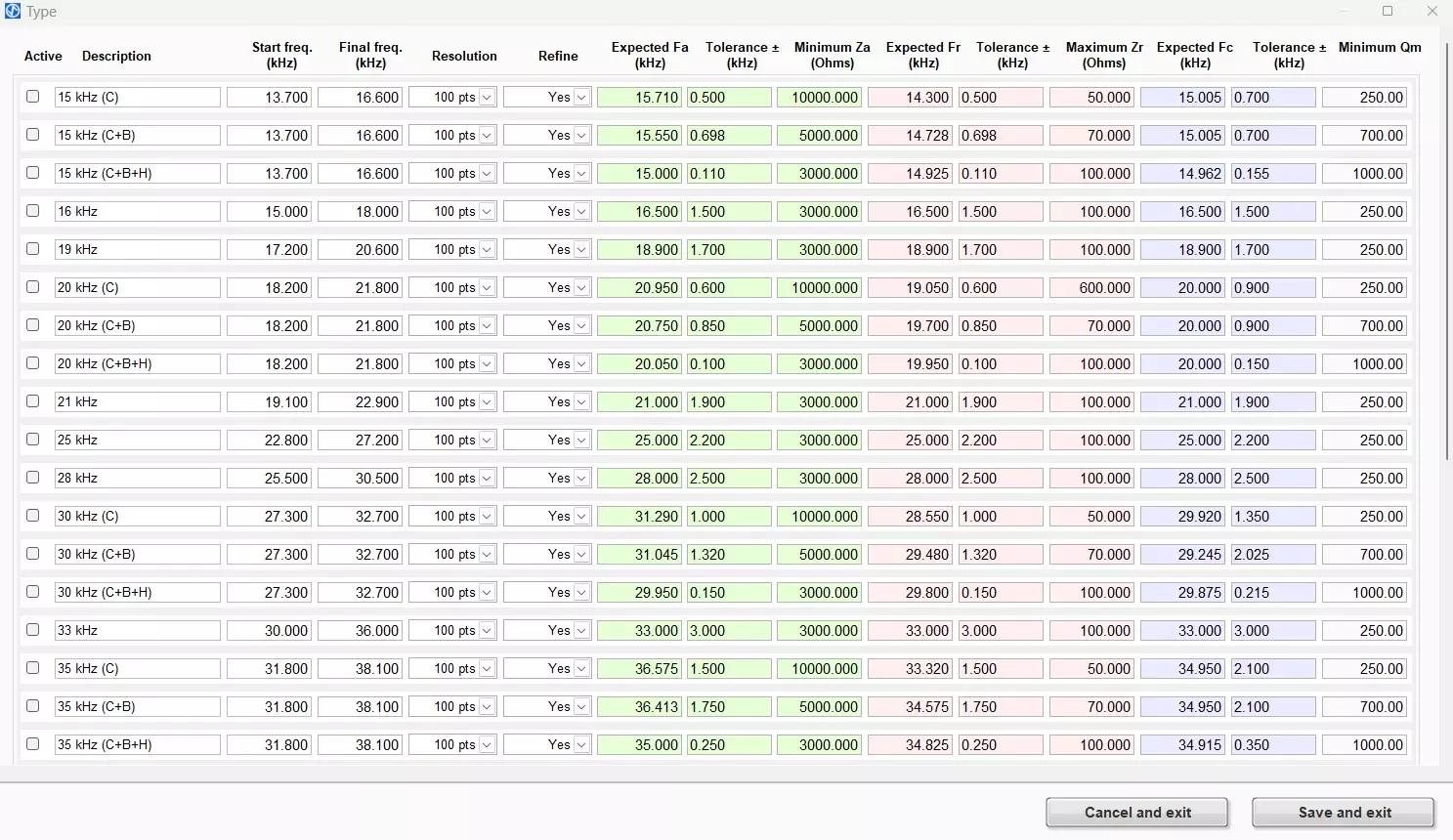
TRZ® Software interface for entering measurement parameters and judgment criteria.
The image below shows an example of a measurement with the results judgment activated. In the “Results” field, in the lower left corner, the green color indicates “pass” and the red one “fail.” The element tested in the example was a medical phacoemulsification transducer. The TRZ® Software also allows superimposing the measurement curve with a reference and with previous measurements with the “Reference” and “Memory” functions.
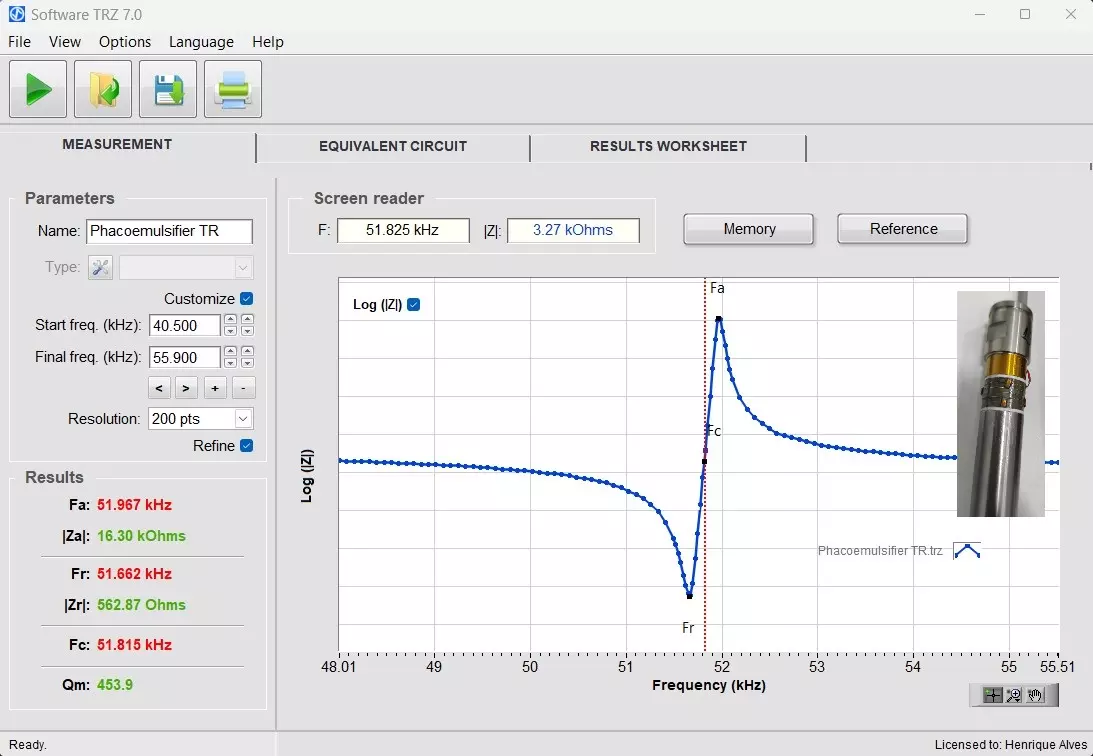
TRZ® Software interface for testing and presentation of results. The superimposed image is of a medical application transducer.
The image below shows the TRZ® Analyzer results interface. Data from this table can be exported to csv file, compatible with MS Excel.
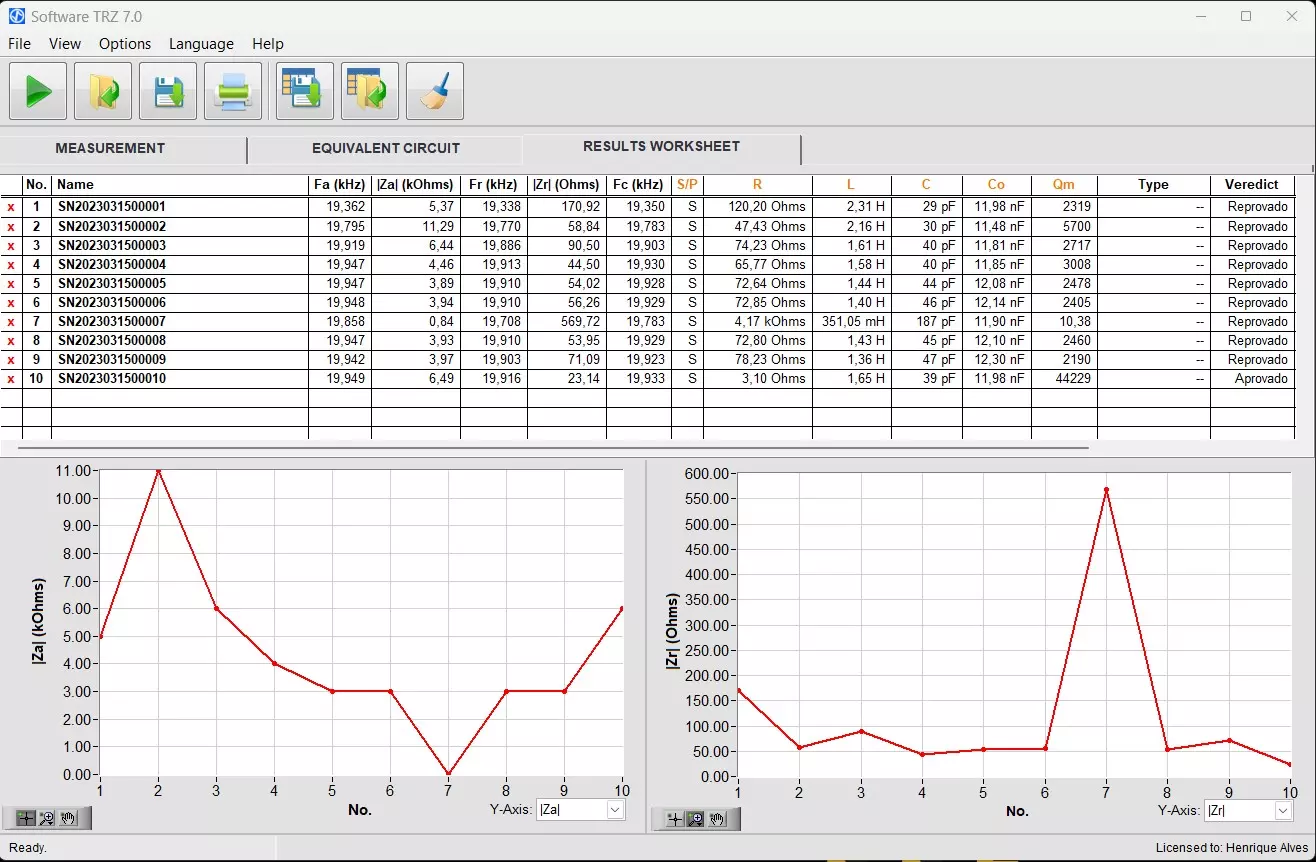
TRZ® Software interface for monitoring and displaying the results of various measurements. It is possible to export data from this table to csv file.
The image below shows an example of a report generated by the TRZ® Analyzer for a medical ultrasonic transducer used in lithotripsy. This report was exported by the TRZ® Software in PDF format. Report generation is an important and useful function of the TRZ® Analyzer Software for traceability in quality control processes.
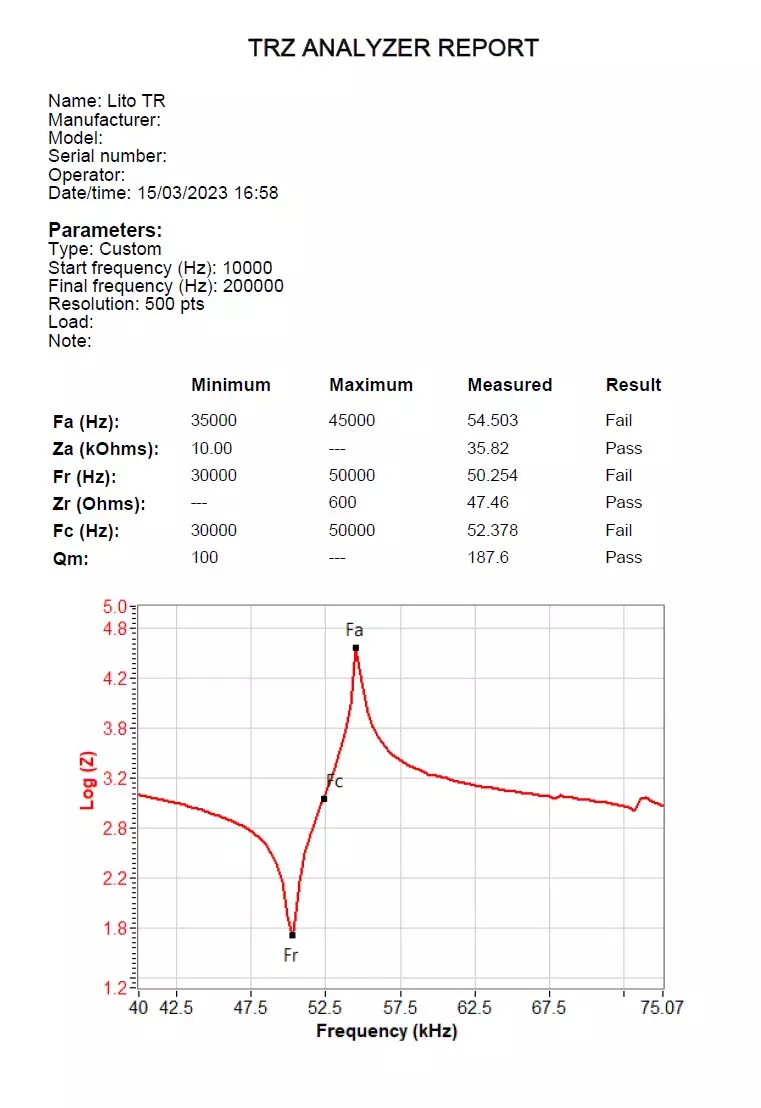
Example of a report generated by the TRZ® Software in PDF format.
The TRZ® Analyzer can test any ultrasonic power element, for medical or industrial application, in the range of 1 to 200 kHz.
